By Sunil Ramkali, Account Director, Life Science & Pharma

You don’t always get out, what you put in
According to the EFPIA The Pharmaceutical Industry in Figures Key Data 2022 report. By the time a medicinal product reaches the market, an average of 12–13 years will have elapsed since the first synthesis of the new active substance.
The same report states that on average, only 1–2 of every 10,000 substances synthesized in laboratories will successfully pass all stages of development required to become a marketable medicine, at a cost between $2–2.5 billion dollars (US).
In 2019, the pharmaceutical industry spent $83 billion dollars (US) on R&D. This is 10 times what the industry spent per year in the 1980s.

This increase in R&D spending can be attributed to several factors, such as the increased need to better understand rare diseases, so we can better address clinical unmet needs, and increased development costs to ensure new treatments address market access barriers by delivering clinically relevant outcomes at an affordable price.
These factors mean that the pharmaceutical industry is having to take more & more risks to bring new treatments to the market. For example, performing large multinational clinical trials, when the outcomes are not certain – uncertainty of improved clinical efficacy versus placebo and / or a clinical comparator.
In 2021, there were several high-profile clinical failures. For example, Johnson & Johnson’s reported disappointing results with its Ad26.Mos4.HIV vaccine. In a phase 2b study that enrolled 2,600 women in southern Africa who were at very high risk of HIV infection. The vaccine was unable to meet the primary endpoint of reducing transmission of HIV by 50% compared to placebo. Resulting in the trial being discontinued.
So, what’s next?
With increasing R&D costs and drug failures in clinical development. Where does that leave the pharmaceutical industry and how can we continue to fund drug development?
More and more pharmaceutical manufacturers are turning to their mature or established brands as a key source of future revenue.
Despite patent expiry or loss of exclusivity of a pharmaceutical brand, there are many markets where healthcare professionals will continue to prescribe the original brand over generic versions, and where patients are prepared to pay extra for the original brand. It is in these brand loyal markets where pharmaceutical manufacturers are continuing to invest in these ‘cash cow’ products to maintain turnover and profitability.
The term ‘cash cow’ originates from the Boston Consulting Group’s growth matrix. Cash Cow is one of the four categories and refers to a product or a business, that will continue deliver turnover / profit throughout its life. Often a cash cow is a product, that has a strong potential to deliver a positive return in a low-growth market.
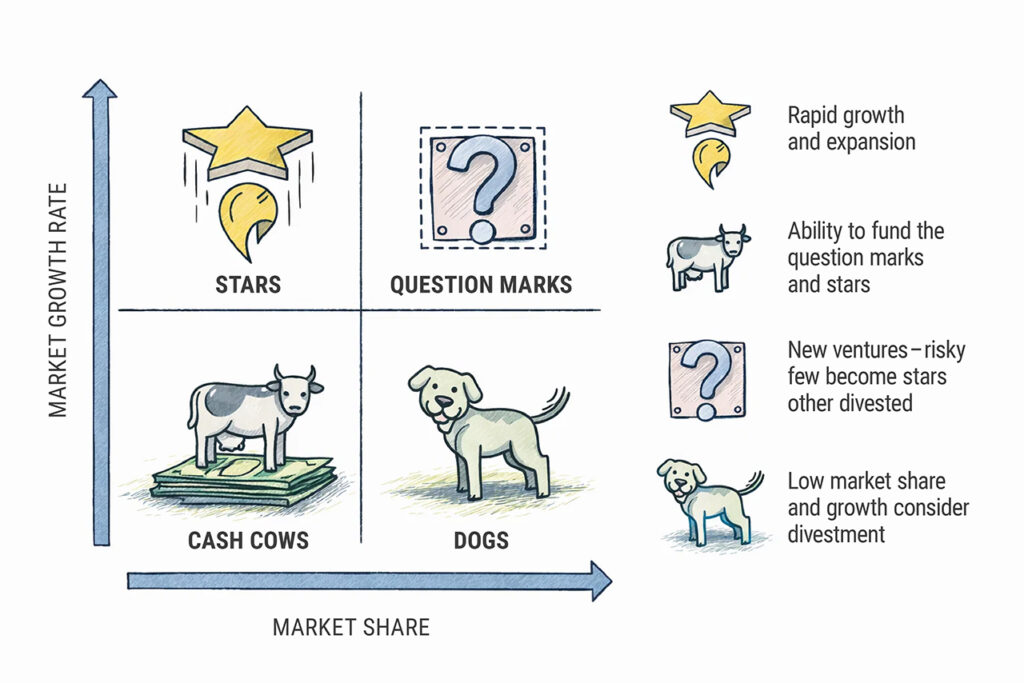
The size of the opportunity
With a population of over 214 million, Brazil’s pharmaceutical market continues to be an attractive market for pharmaceutical manufacturers. According to a report Capturing the Brazilian pharma opportunity from McKinsey & Company, 40% of physicians serving the middle class perceive branded medications to be more effective and appropriate for their patients .
In the same report, 63% of middle-class consumers consider brands very relevant for medicine and would pay a premium for trustworthy ones. The middle-class in Brazil represents approximately one-third of the country’s population, i.e., over 64 million consumers. A figure similar to that of the UK population!
So, what if we could tap into this brand loyalty and convince physicians in key markets to remain loyal to the original brand, despite the availability of generic alternatives?
How not to commercialize a mature brand!
When a pharmaceutical brand is approaching patent expiry or when the patent has expired, there a several commercial initiatives that can be deployed to maximize the turnover and profitable of that brand in these so-called brand loyal markets.
The timing of such commercial initiatives is crucial. Some pharmaceutical manufacturers have had their ‘fingers burnt’ in the past, by implementing such initiatives on the verge of patient expiry, resulting in the initiative being seen as one based on pure greed to maximize profits at the expense of any benefit to the patient.
This was an issue that AstraZeneca experienced when they discontinued Losec® capsules, which at the time was their largest selling drug at that time and was approaching patent expiry. AZ deployed a strategy to discontinue the capsule formulation of Losec and replace it with a tablet formulation of the same brand. In effect, forcing the markets to prescribe the tablet formulation, whose sales would not be affected by the launch of generic versions of the capsule formulation. This strategy created significant backlash in the UK, resulting in AstraZeneca having to restart manufacturing of Losec capsules.
The introduction of the tablets did offer improved clinical benefits over the capsules, in terms of formulation and administration. However, the timing and the nature of the introduction of the tablet formulation created significant PR issues for AstraZeneca.
If AstraZeneca had introduced the Losec tablets well before the patent expiry of the capsules formulation and left the prescribing decision to healthcare professionals / heath authorities on which formulation was suitable for their patients. AZ might have seen a very different outcome to the introduction of Losec tablets and the total revenue of the brand.
So, what can be done – enhanced brand communications
Often pharmaceutical manufacturers will use their therapeutic expertise and develop a drug to supersede a brand that is set to reach patent expiry within the same therapeutic area, e.g., Losec and Nexium® (Gastrointestinal – AstraZeneca), Lexapro® and Brintellix® (Major Depressive Disorders – Lundbeck) & Humira® and Rinvoq® and (AbbVie – Crohn’s disease).
If a manufacturer is unable to replace a brand with a successor within the same therapeutic area. What else can a manufacturer do to maximize the profitability of a brand that has or is about to reach patent expiry?
If we were to rank in order of importance the different commercial initiatives to maximize profitability or the return in investment of a mature brand. Increasing brand value through enhanced product messaging and marketing activities are more likely to result in improved profitability, compared to other activities, such reducing the price to compete with generic versions of the brand. In some situations, it may be a combination of several commercial initiatives, e.g., creating a ‘level playing field’ with generic products on price and then delivering differentiation through effective brand communications.
Even if a brand has been on the market for many years, the opportunity to sharpen or enhance brand messaging will always be there. We are not advocating a change in the brand strategy (if it is working!), but can we deliver and communicate our brand positioning in a more compelling manner and can we create new key messages to reflect this stage of the brand life cycle.
Could we also offer add value services to differentiate the manufacturer and/or the brand, especially in markets where is can be difficult to compete on price, and more holistic approach is required, i.e., a ‘beyond the pill’ strategy.
Of course we need to be aware that the marketing budget available for a mature brand is often low compared to a prioritized brand. Nevertheless, with a smart and creative approach, the ROI for a mature brand can be high.

At W Communication Agency, we have in-depth knowledge of maximizing the commercial value of a mature pharmaceutical brand by strengthening the brand positioning within brand loyal markets through effective product messaging.
If you would like to know more about the possible commercial initiatives to maximize customer adoption of a mature pharmaceutical brand. Why not give me a call or send me an email?
Sunil Ramkali, CEO, W Communication Agency.

Sunil Ramkali
CEO & Account Director, Life Science, W Communication Agency
With over 35 years of experience in global and local sales, marketing, and market access/pricing. Sunil brings a wealth of knowledge to his clients. His insights have been published in numerous articles, including PharmaVoice, and he lectures at Lund University on “Commercial considerations in early drug development”. If you’re seeking guidance in the pharmaceutical industry, Sunil’s expertise can help you achieve success.